Post-mortem Changes and Assessment of Quality
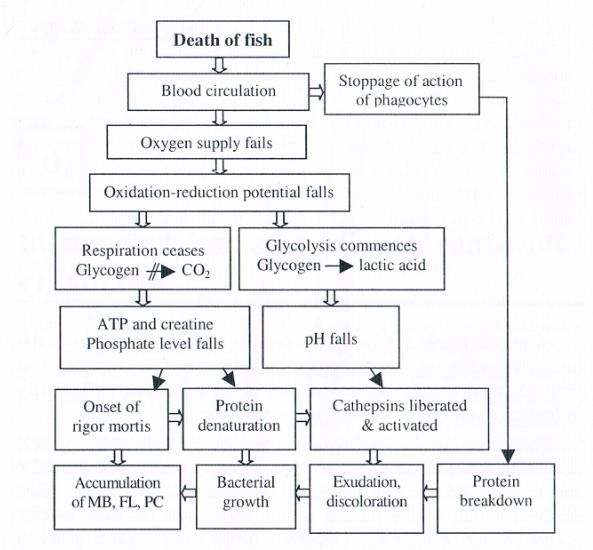
After the death of a fish, considerable changes take place in the body. The changes, however, vary on the basis of size and types of fish, physical condition, season and death condition. Generally following changes are occurred in the carcass of fish.
Following the death of fish, the brain control as well as blood circulation ceases. This results in an inability to resynthesize ATP and to transport various materials essential in living cells. The death-stiffening of the muscle tissue, referred to as rigor mortis, occurs sooner or later and glycolysis forms some organic acids to decrease in pH values.
After finishing the rigor mortis, the muscle tissue loses the stiffness, followed by autolysis forming amino acids and other low molecular weight compounds. Then, microorganisms grow by utilizing these compounds which exist before and after the autolysis, and subsequently, attack high molecular weight compounds. During the spoilage by microorganisms, some specific putrefactive substances such as trimethylamine and histamine are formed depending on fish species. An outline on the postmortem changes in fish and shell fish is given in .
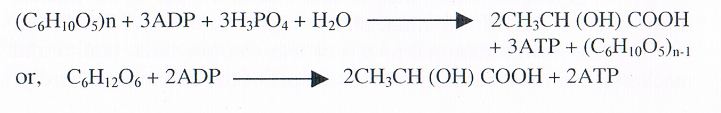
The major source of ATP supply to the muscle fibres is lost after death of fish, since glycogen can no longer be oxidized completely to CO and l-120. Anaerobic glycolysis produces 3 moles (or 2 moles) of ATP for each hexose unit broken down:
For some time after death, ATP is maintained at a definite level in the muscle by active creatine kinase in addition to the above ATP producing reaction:
pH values and glycogen content of animal tissue (Sakaguchi, 1994).
| Species | Glycogen content (G/lOO g wet wt.) |
Ultimate pH |
| Beef | 1.0 | 5.6 |
| Skip-jack (bonito), tuna | 1.2 - 1.4 | 5.6 – 5.7 |
| White fleshed fish | 0.61 – 0.85 | 6.2 – 6.9 |
Mechanism of ATP degradation
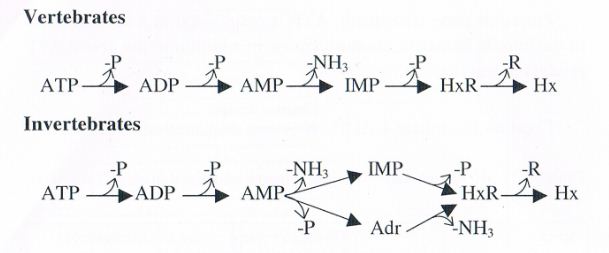
The pathways o ATP breakdown in both vertebrates and invertebrates are shown below. Various steps involved arc carried out by different enzymes present in the fish tissues as well as in bacteria. Hypoxanthin is formed by the action of tissue enzymes and later by bacterial enzymes. The values of hypoxanthin progressively increase from near zero in fresh fish to Levels as high as 8 micro moles/g when the fish is considered spoiled. Hence the values of hypoxanthin can be considered as the measure of both autolytic deterioration and bacterial spoilage. The values of hypoxanthin and freshness of fish correlate well with flavour changes in the period of storage. It is, therefore, considered as a useful index of freshness provided the limits of acceptability are known for a particular species of fish. However, sometimes in certain species very low hypoxaflthin values are observed despite the progress and onset of spoilage. This is because in those species hypoxanthin is immediately broken down into uric acid due to high degree of xanthin Oxidase activity, in certain other species, the breakdown of IMP to hypoxanthin is slow due to reduced activity of some enzymes.
Post-mortem bacteriological changes
Fish generally harbours a large population of bacteria on skin, surface, gills and in the intestine, particularly in case of cultured fish. Usually the bacterial load is in the range of l03 to l05/cm2 in the case of skin surface, l04 to 106/g tissue in the case of gills and 105 to l08/g of the gut contents. When the fish is dead, these bacteria enter the body, proliferate and cause deterioration of muscles. The multiplication of bacteria in fish is controlled by either icing or freezing methods. During freezing cold sensitive bacteria die out and the surviving cold tolerant bacteria take longer time to multiply. Therefore, the rate of bacterial spoilage is slowed down. Usually, a self life extension of 8-15 days is obtained if icing is done properly.
Freezing of fish causes death of bacteria to the extent of 60 to 90% depending on the initial bacterial count. The survivors are mainly gram positive bacteria which can not grow and cause spoilage of frozen fish. 1-lowever, upon thawing at ambient temperature, these bacteria can multiply and cause spoilage of thawed fish.
In post-rigor stage, bacterial spoilage becomes very fast. The rise of muscle pH from acidic to alkaline range for accumulation of volatile bases like ammonia and trimethylaminc produced by spoilt fish enhances bacterial growth. Trimethylamine is produced by the reduction of trimethylamine oxide by bacterial enzyme as well as by tissue enzymes.
Rigor Mortis in Fish
The catabolic process taking place in a freshly dead animal body lead to stiffening of the muscles is known as rigor mortis. There are 3 steps of rigor mortis progress in fish.
Immediately after death, the fish muscles are totally relaxed. The fish is soft and pliable and the texture is firm and elastic to the touch. At this time, the flesh is said to be pre-rigor condition. It is possible to make the muscle contract by stimulation in this stage, for example by means of an electric shock. Eventually, the muscles begin to stiffen and harden and the whole body becomes inflexible, the fish is said to be in-rigor stage. The muscle will no longer contract when stimulated and it never regains this property. After several hours or days, the muscles gradually begin to soften and become limp again. The fish has already passed through time rigor and is said to be in post-rigor stage.
Changes in fish during rigor mortis
Physical changes
When rigor sets in. the muscle becomes hard and stiff and the whole body becomes inflexible. If the fish is filleted pre-rigor, the muscle can contract freely and the fillet will shorten and have a wrinkled surface. Dark muscle may shrink up to 50-52% while the white muscle up to 15-18% of the original length. After rigor, the muscle tissue returns to a relaxed state. With some experience, it is possible to distinguish between the pre-rigor and post-rigor fish since pie-rigor fish is fully flexible and do not show pressure marks after gentle squeezing.
Chemical changes
The course of main biological process leading to rigor mortis is the same in muscles of great variety of species of fish and homeothermic animals. During the transition from living state to full rigor, fish muscle undergoes a series of biochemical changes. The most important reaction is the turn over of ATP, which proceeds for some hours in the dying muscle at about the same rate as in the resting living muscle. Living muscle cells obtain their biochemical energy (ATP) from respiring mitochondria. This aerobic respi rat ion ceases when blood circulation stops after death. Under this circumstances, the enzymatic machinery of the cell which is geared to maintain a constant level of ATP initially use creatine phosphate (CP), a reservoir of phosphate hound energy normally present in the muscle to generate ATP from ADP. ATP level starts Lo fall when 70% or more of the CP has been degraded.
The major anaerobic sources of biochemical energy supply are via the break down of glycogen which through glycolysis does not only generate ATP but also produces lactate. As a result, the protons that are generated during the glycolysis and hydrolysis of ATP to ADP cause a significant decrease of cellular pH from pH 7.2 to pH 5.5. The glycolysis continues until all glycogen is consumed or glycolytic enzymatic system inactivated at pH 5.4. The change in acidity depends also on the liberation of inorganic phosphate and ammonia due to enzymatic degradation of ATP and on the inherent buffering capacity of the muscles. Biochemical causes of rigor-mortis
Rigor mort is is caused by the bonding of myosin heads to the active centers of thin filaments, the actin. This leads to the formation of a rather rigid structure of inter-connected myofilaments, the actomyosin As the individual muscle fibres contain different quantities of ATP. they do not enter rigor at the same time post mortem. Thus, stiffness in muscle sets in gradually. During the later stages of post-mortem changes, the decomposition of nitrogenous compounds leads to an increase in pH in the flesh. The rate of PH changes depends on temperature.
In live or newly died fish, muscular contraction starts when a nervous impulse sets oil a release of Ca from the sarcoplasmic reticulum to the myofibrils. When the Ca concentration increases at the active enzyme site on the myosin filament, the enzyme ATP- ase is activated. This ATP-asc splits the ATP found between the actin and myosin filaments, causing a release of energy. This energy is used as contractile energy making the actin filament slide in between the myosin filaments in a telescopic fashion, thereby contracting the muscle fibre in living tissue. When the reaction is reversed, i.e., when the Ca is pumped back, the contractile ATP-ase activity stops and the filaments are allowed to slip passively past each other, the muscle is relaxed. ATP is one of the most important component for muscle contraction, functioning both as a fuel for contraction and as a plasticizer in the presence of Mg when the muscle is in relaxed state. During rigor mortis progress, as mentioned elsewhere, myofribillar ATP is absent, whereby the actin and myosin filaments stay interlocked as actomyosin. This is why the muscle becomes rigid and stiff during postmortem rigor mortis.